Review of Type Iv Cracking in Piping Welds Ellis Viswanathan
《1. Introduction》
1. Introduction
Grade 91 (Gr.91) steel is a high-chromium (Cr) ferritic–martensitic structural steel that was originally produced past Oak Ridge National Laboratory in the 1970s, and that has been extensively utilized by the power industries, near notably for thick-section boiler components [one]. This alloy steel has high creep resistance, due to the formation of fine secondary-stage One thousand23C6 and MX particles in the matrix and grain boundaries. However, past studies have established that there are 2 possible mechanisms of premature creep failure, which crusade the formation of type IV cracks along the oestrus-affected-zone (HAZ) that can be observed nether short-term or long-term creep tests. Short-term creep tests are performed at higher temperatures and under high stresses, while long-term creep tests are optimized for longer periods of time with lower temperatures and low stresses. The first mechanism of premature creep failure is the coarsening of Chiliad23Csix at high temperature and loftier stress, which occurs during short-term creep testing [2–four]. M23Cvi is viewed every bit a beneficial phase in the initial microstructure, due to its fine particle size. Notwithstanding, volume percentage increase and particle size coarsening of One thousand23C6 are typically observed during or after heat treatment, changing K23C6 into a secondary phase that is detrimental for creep resistance. The second mechanism is the precipitation of the detrimental Z phase; this occurs under the low temperature and low stress of long-term tests [3,v–7], and "ats" away the fine MX particles.
Decades of research on the outcome of creep failure in Gr.91 steel take involved extensive thermodynamic, kinetic, and crystallographic investigations, most of which have focused on direct simulations of creep failure [3,8,9]. However, very few cardinal investigations accept been conducted on the stabilities of the critical secondary phases, which are direct linked to the creep resistance of this steel, even though computational thermodynamics is widely accepted as an extremely useful means of providing predictions and guidance for the design of new alloys—and especially of steels. To address this issue, a thermodynamic investigation of Gr.91 steel was carried out in order to examine the human relationship between various critical temperatures and the stabilities of the Thousand23C6, MX, and Z phases within the HAZ, which can be used to predict and amend the creep resistance under both brusque-term and long-term applications. This investigation paves the mode for a comprehensive agreement of how to optimize the blend composition to ameliorate the creep resistance.
《ii. Literature review》
2. Literature review
《two.1. Type Iv cracks and creep resistance in Gr.91 steel》
2.one. Type IV cracks and pitter-patter resistance in Gr.91 steel
Creep resistance deposition is considered to exist the chief reason for blazon IV cracks in welded Gr.91 steel, which occurs under repeated circadian loading. These cracks are more often than not observed to occur along the outer edge of the HAZ—more specifically, in the finegrain HAZ (FGHAZ) and intercritical HAZ (ICHAZ) (Fig. one), which are created by the welding process and will be explained in further detail beneath. Various works have been carried out in club to understand the formation mechanism of type IV cracks and to propose a method to suppress these cracks. Still, the verbal mechanism leading to critical failure along the HAZ is still unknown, although many suggestions take been made [10–15]. From a thermodynamic viewpoint, information technology has been observed that secondary phases, such as the M23Chalf-dozen, MX, and Z phases, not merely change the microstructure of the HAZ, only besides direct bear on the alloys' failure under long-term and short-term applications. The formation and stability of secondary phases are determined by the alloy limerick, welding process parameters such as the temperature gradient, and the alloy's application conditions. Therefore, thermodynamic simulations are an effective approach to investigate the formation and stability of the secondary phases, which could lead to the determination of the underlying mechanism of type IV cracks.
《2.2. Microstructure of the HAZ》
2.two. Microstructure of the HAZ
The HAZ is considered to exist the most likely place for premature creep failure, since it has been widely observed to form type 4 cracks. 3 main factors affect the microstructure development of the HAZ and its subzones: ① the pinnacle welding temperatures, ② the Ac1 and Ac3 temperatures (defined beneath), and ③ the germination and dissolution of M23Chalf dozen carbides [15,16]. The MX phase is non considered during the formation of the HAZ because information technology does not dissolve or destabilize during welding [17]. Studies have shown that its changes are much smaller, due to its high thermal stability, minor coarsening, and fine distribution inside the microstructure [17,eighteen]. Therefore, the stage stability and coarsening of M23C6 are the principal focus in almost studies on the HAZ formation.
Fig. one shows the schematic microstructure of the HAZ and the location of its three subzones in Gr.91 steel. After the welding process, Gr.91 steel has been observed to grade iii subzones within the HAZ: the coarse-grain HAZ (CGHAZ), FGHAZ, and ICHAZ. It is widely accustomed that the microstructure of the HAZ is closely related to the two Ac temperatures, Ac1 and Ac3. The Ac1 temperature is defined as the temperature at which austenite begins to grade, whereas the Ac3 temperature is the temperature at which austenite transformation is completed. The first zone next to the weld metal is the CGHAZ, which is located nearly the fusion line. In this zone, the peak temperature during welding is to a higher place Ac3, and is likewise higher than the M23C6 stability temperature region. In the CGHAZ, M23C6 particles on the grain purlieus dissolve into the matrix, which reduces the pinning force, leading to coarsening of the grains. In the FGHAZ, which is next to the CGHAZ, the top temperature is also above the Ac3, merely lies inside the 100023Csix stability temperature region. Therefore, the Grand23C6 particles on the grain boundary cannot dissolve into the matrix. Thus, very fine grain size is observed in the FGHAZ due to the grain purlieus pinning upshot from M23C6.
《Fig. 1》

Fig. 1. Schematic microstructure of the HAZ and its three subzones formed during the welding procedure: the fibroid-grain HAZ (CGHAZ), FGHAZ, and ICHAZ.
The ICHAZ, which is the smallest of the three zones, is located furthest from the fusion line and has a peak temperature between Ac1 and Ac3. This zone differs from the other two zones in that upon cooling, a mixture of fresh martensite and ferrite forms, whereas the CGHAZ and the FGHAZ contain generally martensite upon cooling. Hence, from a thermodynamic perspective, the M23Chalf-dozen stability region and the Ac temperatures are considered to be the critical factors determining the microstructure of the HAZ subzones. The Ac3 temperature determines the boundary location between the CGHAZ and FGHAZ, and the Ac1 temperature determines the boundary location between the ICHAZ and the over-tempered region.
《ii.3. Long-term creep failure》
2.iii. Long-term creep failure
Long-term creep tests of Gr.91 steel are defined as tests of a cloth's creep life expectancy that last for 100 000 h or more, with the preferred stresses for testing being beneath 100 MPa [five,7,18–20]. In these tests, the testing temperature will be as depression every bit 550 °C [5,7]. It has been observed that the Z phase will eventually class and reduce the creep resistance of the material [21]. The Z phase has been particularly studied in the last few decades, and information technology has been establish that its formation can lead to a decrease of the fine MX carbonitrides (M(C,N)) and to the disappearance of niobium (Nb)-rich (NbX) MX phases [22,23], both of which are considered to be benign to the textile'south creep strength within the HAZ. It is believed that the formation of the Z phase inside the steel determines the premature creep resistance harm during longterm operations.
《2.4. Short-term creep failure》
ii.iv. Short-term pitter-patter failure
To investigate the creep resistance mechanism over a short time period, speed-up failure creep tests were designed. For instance, the total pitter-patter life resulting in curt-term failure can vary from 100 to 1000 h at temperatures between 575 and 650 °C [24] and nether loftier stresses betwixt 100 and 200 MPa, depending on the rut-handling atmospheric condition prior to testing [3,24]. The chief observation is that the increment and coarsening of Thousand23C6 particles influence the microstructure in the HAZ, and can lower the pitter-patter resistance.
Many factors have been observed to affect the coarsening of M23C6, including the presence of pitter-patter voids in the HAZ [25–27] and neutron irradiation [28–31]. All the same, the mechanism of the coarsening of Grand23Cvi has been best described based on the Ostwald ripening effect [20,32–35], which is considered to be the final phase of precipitation and is mainly dependent on the working temperature of the cloth. Higher temperatures correspond to a college coarsening charge per unit of Yard23Chalf dozen.
《ii.5. Elements pertaining to Gr.91 secondary phases》
two.v. Elements pertaining to Gr.91 secondary phases
Understanding the composition of the secondary phases and the role of each element in contributing to the germination of secondary phases such as the M23Cvi, MX, and Z phases is necessary in order to written report the different mechanisms of pitter-patter resistance degradation in Gr.91 steel at various operation temperatures. The typical concentration of each element and their tolerances nether the American Society of Mechanical Engineers (ASME) standards [36] in the material are provided in Table 1. The "M" in M23Chalf dozen carbides represents the metals Cr, fe (Atomic number 26), or molybdenum (Mo), while the "M" in MX represents vanadium (V) or Nb. The "X" in MX represents carbon (C) or nitrogen (N), and has been observed to vary throughout the material, making these phases carbides (MC), nitrides (MN), or carbonitrides (M,(C,N)) [37]. The Z stage forms at lower temperatures over the long-term usage of the fabric [5]. Its crystal structure has three unlike sites—(Fe,Cr)(Mo,V, Nb)(N,Va)—where Va stands for vacancies. This newspaper adapts the notation of Cr(V,Nb)North for the Z phase for ease of comprehension, since the Fe and Mo concentrations are extremely low.
In full general, the elements in Gr.91 steel can be grouped into two categories—ferrite (α-Fe) stabilizers and austenite ( -Atomic number 26) stabilizers—which affect both the Ac temperatures and the stability of the secondary phases. α-Iron stabilizers are elements added to the steel alloy that accept a large solubility in the α-Atomic number 26 phase, thereby increasing the alloy's Ac temperatures; in contrast,
-Atomic number 26) stabilizers—which affect both the Ac temperatures and the stability of the secondary phases. α-Iron stabilizers are elements added to the steel alloy that accept a large solubility in the α-Atomic number 26 phase, thereby increasing the alloy's Ac temperatures; in contrast,  -Atomic number 26 stabilizers have a big solubility in the
-Atomic number 26 stabilizers have a big solubility in the  -Fe phase, thereby decreasing the Ac temperatures. C and N are both
-Fe phase, thereby decreasing the Ac temperatures. C and N are both  -Atomic number 26 stabilizers [38], and are both essential to the stabilities of the MX and M23Chalf dozen precipitates that form and coarsen during the normalization and tempering process of the heat treatment. Cr, which is an α-Iron stabilizer [38], is the most important element in the alloy. At sufficiently large concentrations, Cr can preclude oxidation and corrosion under aqueous atmospheric condition. Cr is the essential chemical element in the creation of the carbide G23C6 that makes up nigh of the secondary phases that form during the initial stages of heat handling. Other precipitates too form, such as the M7C3 (CrsevenCthree) and M2X (Cr2N) precipitates, if changes are made to the composition [38,39]. Mo is an α-Fe stabilizer that provides a solid solution for the Yard23C6 (Mo23C6) carbides [38]. A limited amount needs to be added in gild to prevent an abundance of both Laves phase (Fe,Cr)2(Mo,West) and δ-phase germination [37]. At depression amounts of Cr, K2C (Mo2C) can form [39]. Lastly, V and Nb (which are
-Atomic number 26 stabilizers [38], and are both essential to the stabilities of the MX and M23Chalf dozen precipitates that form and coarsen during the normalization and tempering process of the heat treatment. Cr, which is an α-Iron stabilizer [38], is the most important element in the alloy. At sufficiently large concentrations, Cr can preclude oxidation and corrosion under aqueous atmospheric condition. Cr is the essential chemical element in the creation of the carbide G23C6 that makes up nigh of the secondary phases that form during the initial stages of heat handling. Other precipitates too form, such as the M7C3 (CrsevenCthree) and M2X (Cr2N) precipitates, if changes are made to the composition [38,39]. Mo is an α-Fe stabilizer that provides a solid solution for the Yard23C6 (Mo23C6) carbides [38]. A limited amount needs to be added in gild to prevent an abundance of both Laves phase (Fe,Cr)2(Mo,West) and δ-phase germination [37]. At depression amounts of Cr, K2C (Mo2C) can form [39]. Lastly, V and Nb (which are  -Fe stabilizers [38]) are strong suppliers to the formation of MX, especially in Gr.91 steel. MX precipitates are extremely stable even at temperatures college than the normalization process temperature, which makes them hard to produce but beneficial in improving the creep resistance.
-Fe stabilizers [38]) are strong suppliers to the formation of MX, especially in Gr.91 steel. MX precipitates are extremely stable even at temperatures college than the normalization process temperature, which makes them hard to produce but beneficial in improving the creep resistance.
《3. Thermodynamic investigation》
iii. Thermodynamic investigation
To determine the stability of the critical secondary phases in Gr.91—that is, the M23Chalf dozen, MX, and Z phases—the calculation of phase diagrams (CALPHAD) approach based on computational thermodynamics was utilized by using Thermo-Calc and its corresponding TCFE8 database. The CALPHAD approach was first developed by Kaufman [xl] to model complex multicomponent materials and their corresponding equilibrium phases. The basic concept of CALPHAD is to gather and analyze thermodynamic data from individual phases and use this data to predict the backdrop of materials under a wide range of temperatures, pressures, and compositional atmospheric condition [41–48].
The chemical limerick of the Gr.91-based organization is shown in Tabular array i. Here, the weight percentage of each element nether ASME standards [36] is compared with the composition chosen for the electric current simulations. The alloy consists of more than 13 elements; however, only the most disquisitional elements were selected in the electric current thermodynamic simulations, particularly for the prediction of the stability of the secondary phases. Therefore, an Iron–Cr–C–Five–Nb–Mo–Northward (Gr.91-based organisation) blend was chosen as the base of operations textile for the investigation.
《Table 1》
Table i Chemic limerick from the ASME standard [36] and simulation of the Gr.91-based organization.

《3.1. Isopleth diagrams of the Gr.91-based system》
3.1. Isopleth diagrams of the Gr.91-based organization
Fig. two shows the isopleth diagrams of the Gr.91-based system with changes in the carbon concentration. More than specifically, Fig. 2(a) is the isopleth diagram for a C concentration ranging from 0 to 0.2 wt% at temperatures betwixt 600 and 1600 °C, whereas Fig. 2(b) is the diagram for 0–0.fifteen wt% of C at 700–thousand °C. The isopleth diagrams prove the phase stability region for the α and  matrix phases along with the critical secondary phases, including the MX phase, K23Chalf-dozen phase, and Z phase.
matrix phases along with the critical secondary phases, including the MX phase, K23Chalf-dozen phase, and Z phase.
《Fig. two》

Fig. 2. Phase diagrams of the Gr.91-based system, with the Ac1 (blue) and Ac3 (red) temperatures highlighted. (a) Overall phase diagram of the Gr.91-based system for temperature ranges of 600–1600 °C at a range of 0–0.2 wt% of C; (b) close-upwards of the stage diagram at temperature ranges of 700–m °C and 0–0.15 wt% of C.
It is worth noting that the MX phase has the same facecentered cubic (fcc) crystal structure as the matrix  stage. Nevertheless, the Gibbs energy of the MX has a miscibility gap, which means that ii fcc MX phases with different compositions tin exist formed within a specific temperature region. This finding agrees well with the experimental observations for this particular family unit of alloys [49–53]. A more detailed word on the Gibbs energy of the MX phases and the matrix
stage. Nevertheless, the Gibbs energy of the MX has a miscibility gap, which means that ii fcc MX phases with different compositions tin exist formed within a specific temperature region. This finding agrees well with the experimental observations for this particular family unit of alloys [49–53]. A more detailed word on the Gibbs energy of the MX phases and the matrix  phase is provided in the subsection below. The MX phase can exist described every bit a solid solution phase with two sublattices: (V,Nb)(C,Due north). The get-go sublattice is mainly occupied with Five and Nb, while C and N occupy the second sublattice. In the current work, MX1 is used to draw the MX stage, which has a higher site fraction of N than C in the second sublattice. MX2 is used to describe the phase in which the site fraction of C is higher than that of Due north in the 2nd sublattice.
phase is provided in the subsection below. The MX phase can exist described every bit a solid solution phase with two sublattices: (V,Nb)(C,Due north). The get-go sublattice is mainly occupied with Five and Nb, while C and N occupy the second sublattice. In the current work, MX1 is used to draw the MX stage, which has a higher site fraction of N than C in the second sublattice. MX2 is used to describe the phase in which the site fraction of C is higher than that of Due north in the 2nd sublattice.
Detailed simulations accept been done to testify that the composition of the M23C6 phase—that is, (Cr,Fe,Mo)23C6—does non change much in this steel alloy; this stage has Cr as the ascendant element in the first sublattice, and forms in the lower temperature region [38]. The Z stage—that is, (Iron,Cr)(Mo,V,Nb)(Northward,Va)—forms at even lower temperatures; its limerick does not change much, and Cr and Five are e'er the dominant elements in the first and the second sublattices, respectively [23].
At 0.05 wt% of C, the showtime secondary phase that is precipitated out during the temperature change from 1243 to 896 °C is the MX1 stage. When the temperature drops to the Ac3 temperature (896 °C), the  phase starts to transform into the α phase. Equally the temperature decreases further, the other ii secondary phases—that is, the M23Chalf-dozen phase and Z stage—become stable. It should be noted that in regions with a carbon concentration higher than 0.1 wt%, ii types of MX phases—MX1 and MX2—become stable due to the miscibility gap. For instance, with an increment in carbon concentration, the stable phases change from
phase starts to transform into the α phase. Equally the temperature decreases further, the other ii secondary phases—that is, the M23Chalf-dozen phase and Z stage—become stable. It should be noted that in regions with a carbon concentration higher than 0.1 wt%, ii types of MX phases—MX1 and MX2—become stable due to the miscibility gap. For instance, with an increment in carbon concentration, the stable phases change from  + MX1 to
+ MX1 to  + MX1 + MX2 around 0.155 wt% of C at 1000 °C, as shown in Fig. 2(a).
+ MX1 + MX2 around 0.155 wt% of C at 1000 °C, as shown in Fig. 2(a).
《3.two. Gibbs energy of the fcc structure ( phase and MX phase)》
three.two. Gibbs energy of the fcc structure ( phase and MX stage)
phase and MX stage)
In the CALPHAD arroyo, a single Gibbs energy description is typically used to draw the phases that share the same crystal structure [23]. As mentioned above, the  , MX1, and MX2 phases all have the fcc construction. The MX phase is considered to be beneficial for creep resistance because information technology is thermodynamically more stable and has a very depression coarsening rate in comparison with M23C6 under short-term applications [38]. Therefore, information technology is worthwhile to take a closer look at this stage. Fig. 3 shows the Gibbs energies of the
, MX1, and MX2 phases all have the fcc construction. The MX phase is considered to be beneficial for creep resistance because information technology is thermodynamically more stable and has a very depression coarsening rate in comparison with M23C6 under short-term applications [38]. Therefore, information technology is worthwhile to take a closer look at this stage. Fig. 3 shows the Gibbs energies of the  , MX1, and MX2 phases at grand °C. It can be seen that the Gibbs energies of these three phases are very different, fifty-fifty though the phases share the same crystal construction. In the region with less than 0.15 wt% of C,
, MX1, and MX2 phases at grand °C. It can be seen that the Gibbs energies of these three phases are very different, fifty-fifty though the phases share the same crystal construction. In the region with less than 0.15 wt% of C,  and MX1 are the only stable phases, while MX2 becomes stable in the region with a higher C concentration. This result conspicuously explains the phase transformation shown at 1000 °C in Fig. 2(a).
and MX1 are the only stable phases, while MX2 becomes stable in the region with a higher C concentration. This result conspicuously explains the phase transformation shown at 1000 °C in Fig. 2(a).
《Fig. 3》
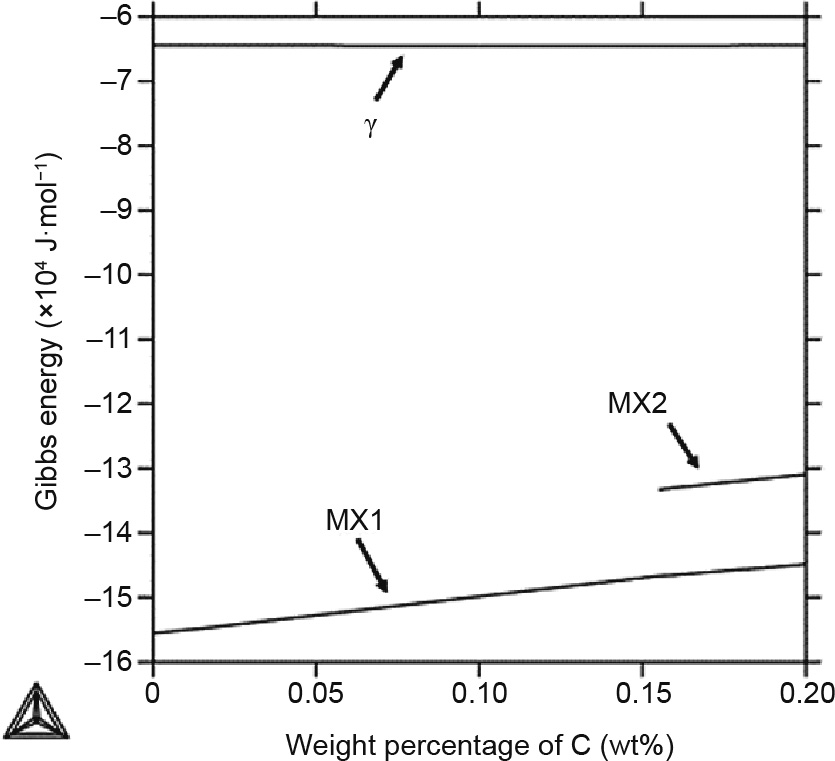
Fig. 3. Gibbs energy of the  , MX1, and MX2 phases in the Gr.91-based arrangement at k °C from 0 to 0.xx wt% of C.
, MX1, and MX2 phases in the Gr.91-based arrangement at k °C from 0 to 0.xx wt% of C.
《iii.3. Ac1 minimum austenite transformation temperature and Ac3 threshold ferrite temperature》
iii.3. Ac1 minimum austenite transformation temperature and Ac3 threshold ferrite temperature
The two Air-conditioning temperatures—Ac1 and Ac3—are critical in understanding the phase stabilities and the microstructure development in the HAZ. In particular, the Ac3 temperature determines the boundary between the FGHAZ and ICHAZ, and the Ac1 temperature determines the purlieus betwixt the ICHAZ and the overtempered region, equally discussed above (Fig. 1). Therefore, these two Ac temperatures are closely linked with the formation of blazon IV cracks, making it worthwhile to investigate the factors that impact them. Fig. 4 shows the change of the Ac1 and Ac3 temperatures in the Gr.91-based system (solid lines) in comparison with a Fe–C binary system (dashed lines).
With an increase in the carbon concentration, equally shown in Fig. 4(a), the Ac1 temperature of the Gr.91-based system drops from 890 to 830 °C, with the biggest divergence happening in the beginning, up to 0.01 wt% of C, and then gradually decreasing afterwards. Similar behavior is seen for the Fe–C binary system; notwithstanding, the sudden alter happens at a slightly higher carbon concentration and drops from 910 to 738 °C. This sudden change in both systems is due to the phase transformation. The Ac1 temperature from the Gr.91-based system is generally much higher than that from the Fe–C system. This major difference indicates that the alloying elements in Gr.91 profoundly increase the Ac1 temperature. Fig. 4(b) shows the Ac3 temperature vs. the carbon concentration. The temperature drops from 954 °C for 0 wt% of C to 850 °C for 0.2 wt% of C for the Gr.91-based organisation, whereas information technology drops from 910 to 837 °C for the Atomic number 26–C system. Similar to Fig. 4(a), the slope change in the curve for the Gr.91-based system at around 0.08 wt% of C represents a phase transformation from α +  + MX1 + MX2 + M23Cvi to α +
+ MX1 + MX2 + M23Cvi to α +  + MX1 + M23C6. A linear relationship is observed for the Fe–C binary organisation, because there is no phase transformation—that is, α and
+ MX1 + M23C6. A linear relationship is observed for the Fe–C binary organisation, because there is no phase transformation—that is, α and  are the only 2 stable phases in this temperature range. Overall, these findings show that Gr.91 shares similar Ac3 temperatures with the Fe–C binary system in the typical C concentration range for Gr.91—that is, for about 0.one wt% of C.
are the only 2 stable phases in this temperature range. Overall, these findings show that Gr.91 shares similar Ac3 temperatures with the Fe–C binary system in the typical C concentration range for Gr.91—that is, for about 0.one wt% of C.
《Fig. 4》
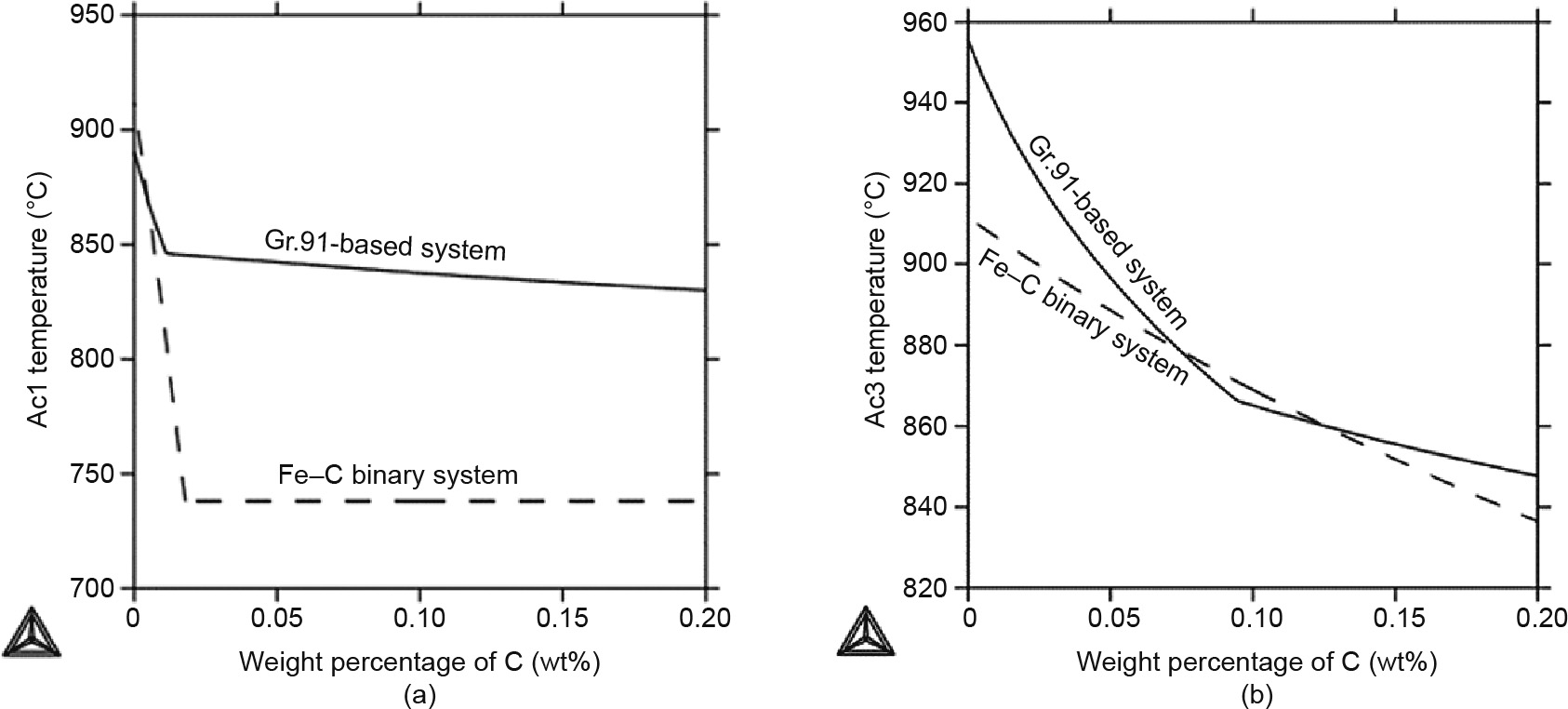
Fig. iv. (a) Ac1 and (b) Ac3 temperatures (°C) vs. weight per centum of C for the Gr.91-based organisation simulation (solid line) in comparison with the Air conditioning temperatures of the Iron–C binary system (dashed line).
《3.4. Mole/site fraction of secondary phases under the equilibrium condition》
3.4. Mole/site fraction of secondary phases nether the equilibrium condition
Fig. 5 shows the mole fraction of the secondary phases—that is, the M23C6, MX, and Z phases—in the temperature range between 600 and 1400 °C for the Gr.91-based steel. It can be seen that the MX1 phase is stable between 770 and 1260 °C, while a very limited amount of the fcc MX2 phase shows upwards briefly between 847 and 890 °C. Fig. 6 shows the changes in the site fraction in the get-go and 2d sublattices for the MX1 and MX2 phases. For MX1, it shows that the dominant species in the beginning sublattice changes from Nb to V with the subtract in temperature, while North is always the dominant species in the second sublattice. For the MX2 phase, Nb is e'er the dominant species for the outset sublattice, while C has a slightly college site fraction than N in the second sublattice. Therefore, V is an important element in MX1 at lower temperatures.
《Fig. 5》
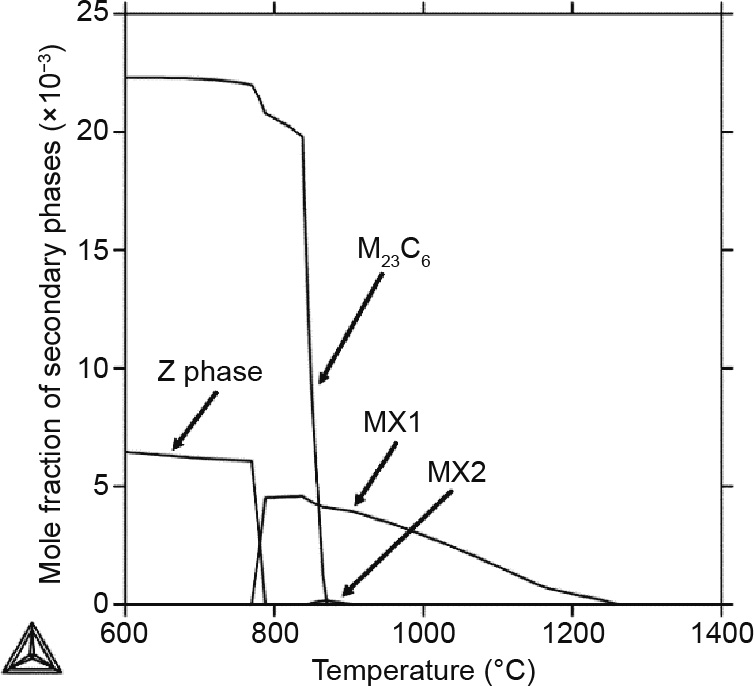
Fig. five. Mole fraction of the secondary phases in the Gr.91-based system.
《Fig. 6》

Fig. 6. Site fraction of Nb, V, C, and N (a,b) the MX1 and (c,d) the MX2 phases of the Gr.91-based arrangement.
M23C6 becomes stable when the temperature is lower than 870 °C, and chop-chop becomes the dominant secondary phase with a decrease in temperature. Its dramatic concentration increment is due to the  to α matrix stage transformation in that temperature range, which corresponds to the sudden carbon solubility subtract in the matrix.
to α matrix stage transformation in that temperature range, which corresponds to the sudden carbon solubility subtract in the matrix.
Fig. five also shows that the Z phase is stable at temperatures below 790 °C, when MX1 becomes unstable. The formation of the Z phase takes Five from the MX1 phase and causes the decomposition of the MX1 phase. This likely to exist the reason why information technology has been experimentally observed that the Z phase "eats" away the MX phase during depression-temperature long-term creep tests [54].
《three.v. Equilibrium cooling and Scheil simulations》
3.v. Equilibrium cooling and Scheil simulations
The in a higher place equilibrium cooling simulation can be used to empathize the phase stability change during the ideal wearisome cooling. However, a existent sample does not ever reach equilibrium at each temperature under typical cooling atmospheric condition. Therefore, both equilibrium cooling and Scheil simulations were carried out in society to obtain a bones understanding of the phase stabilities under existent cooling conditions.
The equilibrium simulation, shown in Fig. 5, predicts the germination of secondary phases under equilibrium cooling weather condition— that is, the sample reaches global equilibrium at each temperature. It is used to simulate the stage stabilities for an extremely ho-hum cooling rate. Meanwhile, the Scheil simulation predicts the formation of secondary phases under conditions similar to the quench condition. At that place are 3 major assumptions for Scheil simulations: ① No diffusion occurs in solid phases in one case they are formed; ② the liquid phase is homogeneous all the time; and ③ the local equilibrium exists at the interface of the liquid and solid phases. It has been widely accepted that the equilibrium cooling and Scheil simulations can be adopted as the two boundary weather. In other words, equilibrium cooling corresponds to an extremely slow cooling rate, while the Scheil simulation corresponds to a quenching procedure. Therefore, the quantity of the solid phases nether existent weather condition should fall between what is predicted by the equilibrium cooling and Scheil simulations. In addition, using these two simulations may provide guidance regarding stage stability changes under various processing weather, such as normalization, tempering, welding, and post-welding heat treatment. Under these conditions, the phase stability of the arrangement will change toward the equilibrium status as the time increases.
Fig. 7 shows the formation of the secondary phases—the M23C6, MX1, Z, and MX2 phases—during the solidification process of the Gr.91-based steel from the equilibrium cooling (solid line) and Scheil (dashed line) simulations. Equally shown in Fig. 7(a), the Thou23C6 phase precipitates out at 870 °C under equilibrium cooling conditions, but forms at 1225 °C during the Scheil simulation. In addition, much less of the 100023C6 phase will class during the Scheil simulation than during equilibrium cooling. This result indicates that the concentration of the M23C6 phase formed during the existent cooling process may exist less than that formed during the equilibrium simulations. Therefore, Gr.91 steel has a tendency to form much more of the M23Cvi phase nether annealing/performance weather condition at elevated temperatures. This finding explains very well why much more of the M23C6 phase has been observed in Gr.91 samples nether high-temperature short-term creep testing weather [24].
On the other paw, the MX1 phase volition form at 1260 °C during equilibrium cooling, as shown in Fig. seven(b), but volition disappear at temperatures below 770 °C. Under fast cooling weather, the MX1 stage will form at 1410 °C; however, its maximum content is much lower than that resulting from equilibrium cooling. This finding indicates that the observed MX phase is a metastable phase at typical operation temperatures, that its formation is due to the fast cooling speed, and that it has a trend to disappear under high-temperature short-term creep testing conditions. The observations in Figs. 7(a) and (b) explain very well the increased molar volume of the Thousand23Chalf dozen phase and the disappearance of the MX phase nether high-temperature short-term creep exam atmospheric condition [3].
Furthermore, Fig. seven(c) shows that the Z phase is a stable stage under equilibrium cooling atmospheric condition, and its formation temperature is lower than those of the Chiliad23C6 and MX phases. Yet, the Z phase does non form during the Scheil simulation, indicating that formation of the Z stage is prohibited by the fast cooling rate. This finding explains very well why the Z phase is non observed in the initial microstructure, and why it gradually shows upwards and eats abroad the MX phase nether low-temperature long-term creep test conditions [54]. For the MX2 phase, only a small fraction forms during equilibrium simulations in a short temperature range, as shown in Fig. two(b) and Fig. 5, while the MX2 phase is not stable at all during the Scheil simulations. Its stability volition greatly depend on the cooling rate and alloy compositions.
《Fig. 7》
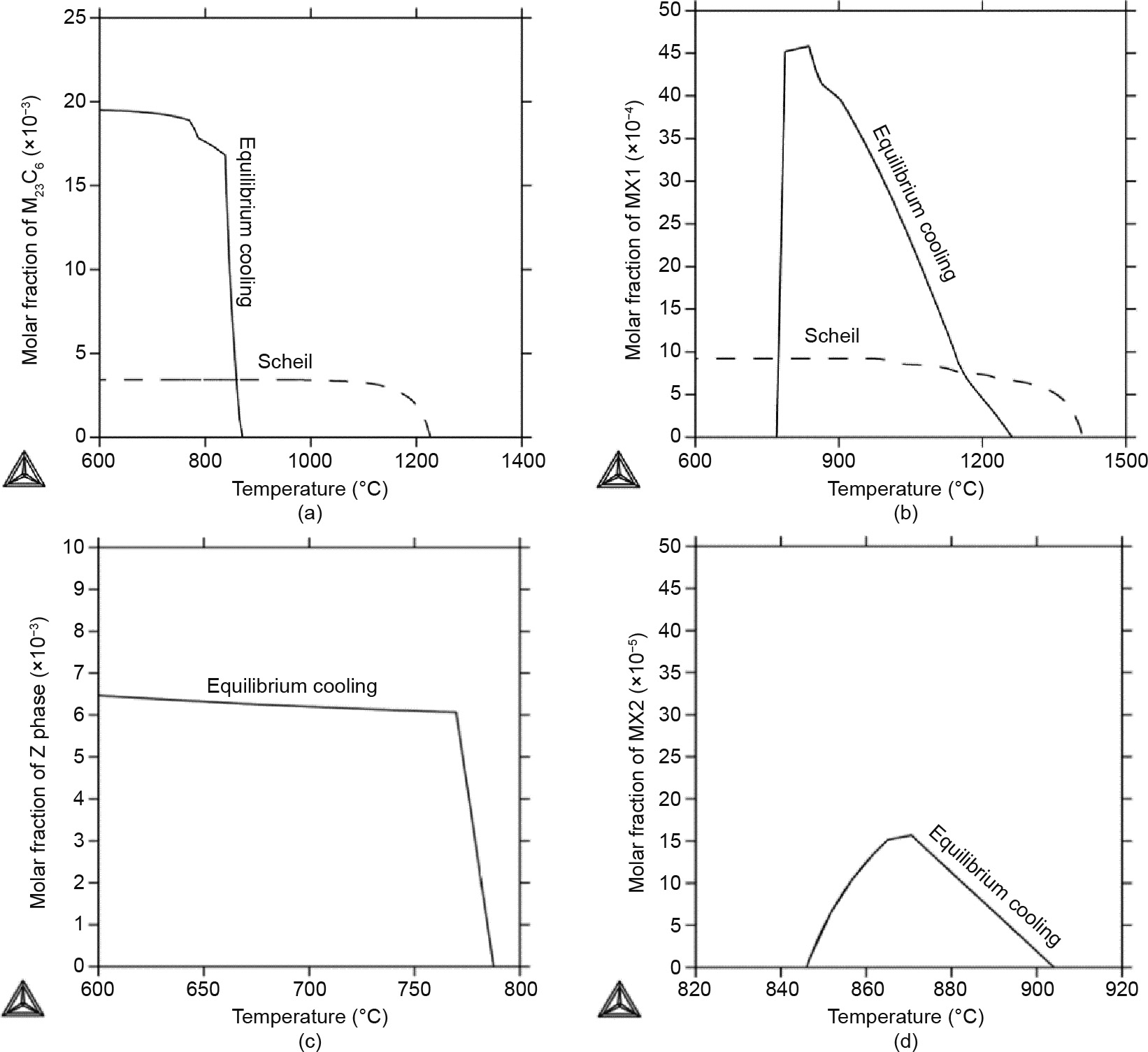
Fig. 7. Results of the equilibrium cooling (solid line) and Scheil (dashed line) simulations for the Gr.91-based system. Molar fractions for (a) the M23C6 stage, (b) the MX1 phase, (c) the Z stage, and (d) the MX2 phase. The Z phase and MX2 stage were not stable during the Scheil simulations.
《iii.six. Alloy composition effects on creep resistance》
iii.half-dozen. Alloy limerick effects on creep resistance
Based on the systematic thermodynamic simulations, it was found that the stabilities of various secondary phases are imperative to the creep resistance of Gr.91 alloys. The simulation results too show that the alloy composition affects the stabilities of the secondary phases. To improve the creep resistance of Gr.91 alloys, the alloy composition needs to exist optimized in order to obtain the desired microstructure in the HAZ later on the welding process.
As shown in Fig. 8, at that place are four unlike critical temperatures for Gr.91 alloys. Ac1 (the blueish line) is the purlieus between the ICHAZ and the over-tempered region, while Ac3 (the carmine line) is the purlieus betwixt the FGHAZ and the ICHAZ during welding. The G23Chalf-dozen threshold temperature (the black solid line) is defined as the maximum temperature at which M23C6 is thermodynamically stable. In welded alloys, this corresponds to the boundary between the FGHAZ and the CGHAZ. Equally mentioned above, type Four cracks only occur within the FGHAZ and ICHAZ [55]. Many suggestions have been fabricated to explain why it is possible for type IV cracks to be located within this region of the material [3,15]; however, from a thermodynamic bespeak of view, ii secondary phases—M23C6 coarsening and Z phase formation—take been observed and theorized to decrease the pitter-patter resistance between the FGHAZ and the ICHAZ [20,23,56].
《Fig. 8》

Fig. 8. Gr.91 M23C6 (black solid line) and Z phase (dashed line) threshold diagram, along with the Ac1 (bluish solid line) and Ac3 (crimson solid line) temperatures.
Bold that the temperature gradient inside the HAZ during the welding process is not changing, adjusting the Ac1, Ac3, and Yard23Cvi threshold temperature will have a big touch on the thickness of the FGHAZ and ICHAZ. To reduce the whole thickness of the FGHAZ and ICHAZ, it is necessary to reduce the temperature difference between the Ac1 and 100023C6 threshold temperatures. As shown in Fig. 8, reducing the carbon concentration of Gr.91 should exist helpful in reducing the risk of type Iv nifty.
High-temperature brusk-term pitter-patter resistance is related to the coarsening of the M23C6 phase [20,32–35]. Therefore, the creep resistance tin be improved by reducing the thermodynamic stability of Yard23Csix. Based on Fig. 8, the Chiliad23C6 threshold temperature will greatly decrease with a decrease in carbon concentration, which will decrease the stability of the M23C6 at high temperatures and thereby increase the high-temperature short-term creep resistance.
Depression-temperature long-term creep resistance is related to the formation of the detrimental Z phase. Based on previous observations, the North-rich MX phase will eventually disappear through Cr diffusion from the grain matrix into MX, eventually transforming the MX stage into the Z phase [23]. From this observation, the assumption is that by reducing the Z phase threshold temperature, which is the maximum temperature at which the Z phase is thermodynamically stable, information technology should exist possible to profoundly improve the long-term creep resistance. As illustrated in Fig. 8, a higher carbon concentration will be helpful in improving the short-term creep resistance.
Similar thermodynamic simulations could exist performed for the alloying elements, in order to investigate the effects of alloy composition on pitter-patter resistance. The approach used in the current piece of work paves the way for the blend composition optimization of Gr.91 in order to improve the creep resistance of this alloy under various applications.
《4. Summary and time to come work》
four. Summary and future work
This piece of work establishes a fundamental thermodynamic agreement using the CALPHAD approach by analyzing and simulating the relationship amidst the Ac temperatures, M23Csix, and Z stage threshold temperatures, and stability and compositional change of the M23C6, MX, and Z phases in the HAZ of Gr.91 steel.
Two different thermodynamic creep mechanisms nether dissimilar application atmospheric condition contributing to a decrease in creep resistance and, eventually, type Four cracks in the HAZ were discussed: ① Thou23Chalf-dozen coarsening under low stress and loftier temperatures for curt-term testing conditions through the Ostwald ripening effect [20,32–35]; and ② Z phase formation under high stress and low temperatures for long-term application conditions, which eventually decreases the overall MX phase stability within Gr.91 [22,23].
Equilibrium cooling and Scheil simulations were carried out to show the stage stabilities under ii farthermost cooling atmospheric condition—that is, extremely slow cooling and quench weather condition. The Scheil simulations showed the formation of a limited amount of the M23Chalf dozen and MX1 phases under fast cooling conditions and in a higher place 1200 °C. In comparison, the equilibrium cooling simulations indicated that much more of the M23C6 and Z phases grade at depression temperatures. Meanwhile, the MX stage is thermodynamically unstable and may disappear at functioning temperatures. This finding successfully explains the experimental observations of the formation of more than Thousand23C6, the formation of the Z stage, and the disappearance of the MX phase under creep exam weather.
In addition, effort was initiated to investigate the elements' effects on creep resistance by using carbon every bit a case study. To be specific, the effect of carbon on the four disquisitional temperatures—that is, Ac1, Ac3, the Thou23Chalf-dozen threshold temperature, and the MX threshold temperature—was investigated. The findings show that the addition of carbon may have various furnishings on the microstructure and phase stabilities. Information technology may increment the overall thickness of the FGHAZ and ICHAZ during the welding process, which in turn may increase the likelihood of type IV cracking. Furthermore, the addition of carbon may increase the stability of K23Chalf-dozen and decrease the stability of the Z phase, which may be detrimental to the shortterm creep resistance but benign to the long-term creep resistance. Yet, based on the thermodynamic model of the secondary phases—that is, the M23Csix and Z phases—in Gr.91, information technology cannot be ended that there is a specific carbon concentration that tin can alter the overall stability of the secondary phases. The model highlights the overall trend of the threshold stage stability, while the mechanical properties can exist contradistinct depending on the welding weather condition, which can in turn affect the HAZ microstructure. Therefore, the alloy composition needs to be optimized through both specific heat treatment atmospheric condition and creep experiments. In improver, similar simulations tin can reveal the function of various alloying elements in type IV great in the HAZ and in the pitter-patter resistance of Gr.91 steel, and volition be carried out in hereafter studies.
《Acknowledgements》
Acknowledgements
This material is based upon work supported by the United states of america Department of Energy (DOE) (DE-FE0027800). The authors would like to thank the DOE National Energy Applied science Laboratory programme managers, Dr. Karol Schrems and Dr. Jessica Mullen, and Dr. Wei Zhang from Ohio State University, for their back up and guidance.
《Disclaimer》
Disclaimer
This paper was prepared as an account of work sponsored by an agency of the United states government. Neither the Usa regime nor any bureau thereof, nor whatever of their employees, makes any warranty, limited or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any data, apparatus, product, or procedure disclosed, or represents that its use would non infringe upon privately owned rights. Reference herein to whatsoever specific commercial product, process, or service by merchandise name, trademark, manufacturer, or otherwise does non necessarily constitute or imply its endorsement, recommendation, or favoring past the United states of america government or any bureau thereof. The views and opinions of the authors expressed herein do not necessarily state or reverberate those of the United States government or any agency thereof.
《Compliance with ethics guidelines》
Compliance with ethics guidelines
Andrew Smith, Mohammad Asadikiya, Mei Yang, Jiuhua Chen, and Yu Zhong declare that they have no conflict of interest or financial conflicts to disclose.
References
[1] Abe F, Okada H, Wanikawa S, Tabuchi M, Itagaki T, Kimura K, et al. Guiding principles for development of advanced ferritic steels for 650 C USC boilers. In: Proceedings of the Seventh Liege Conference on Materials for Advanced Power Engineering; 2002 Sep 30–Oct 2; Liege, Kingdom of belgium; 2002. p. 1397–406. link1
[2] Bhadeshia HKDH. Design of ferritic pitter-patter-resistant steels. ISIJ Int 2001;41 (6):626–40. link1
[iii] Abson DJ, Rothwell JS. Review of blazon IV smashing of weldments in 9–12%Cr creep strength enhanced ferritic steels. Int Mater Rev 2013;58(8):437–73. link1
[4] Abe F. Coarsening behavior of lath and its event on creep rates in tempered martensitic 9Cr–Westward steels. Mater Sci Eng A 2004;387–389:565–9. link1
[5] Sawada K, Kushima H, Kimura Yard, Tabuchi Yard. Z-stage formation and its effect on long-term creep strength in 9–12%Cr creep resistant steels. Trans Indian Inst Met 2010;63(two–3):117–22. link1
[six] Hald J. Materials for advanced power engineering. In: Proceedings of the 8th Liege Conference on Materials for Advanced Ability Applied science; 2006 Sep 18– 20; Liege, Belgium; 2006. link1
[seven] Kimura K, Sawada One thousand, Kushima H, Toda Y. Influence of chemical composition and heat treatment on long-term creep strength of Grade 91 steel. Procedia Eng 2013;55:2–9. link1
[8] Abd El-Azim ME, Nasreldin AM, Zies G, Klenk A. Microstructural instability of a welded joint in P91 steel during creep at 600 C. Mater Sci Technol 2005;21(7):779–ninety. link1
[ix] Abe F, Taneike Thou, Sawada K. Alloy design of creep resistant 9Cr steel using a dispersion of nano-sized carbonitrides. Int J Press Vessels Pipage 2007;84(one–ii): 3–12. link1
[10] Gooch DJ, Kimmins ST. A study of type Four bang-up in ane/2% CrMoV/2 1/4% CrMo weldments. In: Proceedings of the Third International Conference on Creep and Fracture of Engineering science Materials and Structures; 1987 Apr 5–10; Swansea, United kingdom; 1987. p. 698–703. link1
[eleven] Kimmins ST, Coleman MC, Smith DJ. An overview of creep failure associated with heat affected zones of ferritic weldments. In: Proceedings of the Fifth International Conference on Pitter-patter and Fracture of Applied science Materials and Structures; 1993 Mar 28–Apr 2; Swansea, Uk; 1993. p. 681–94. link1
[12] Kimmins ST, Smith DJ. On the relaxation of interface stresses during pitter-patter of ferritic steel weldments. J Strain Anal Eng Des 1998;33(three):195–206. link1
[thirteen] Ellis FV, Viswanathan R. Review of type IV cracking in piping welds. In: Proceedings of the International Conference on Integrity of High Temperature Welds; 1998 Nov iii–4; London, U.k.; 1998.
[14] Nishimura N, Iwamoto K, Yamauchi G, Masuyama F, Imamoto T, Yokoyama T. Development of life assessment organization for high energy piping in fossil power boilers. In: Proceedings of the fourth International Conference on Reliability, Maintainability and Safety; 1999 May eighteen; Shanghai, China; 1999. p. 347–52. link1
[15] Francis JA, Mazur W, Bhadeshia HKDH. Review type Four cracking in ferritic power constitute steels. Mater Sci Technol 2006;22(12):1387–95. link1
[16] Mannan SL, Laha Thousand. Creep behavior of Cr–Mo steel weldments. Trans Indian Inst Met 1996;49(4):303–20. link1
[17] Yu X, Babu SS, Terasaki H, Komizo Y, Yamamoto Y, Santella ML. Correlation of precipitate stability to increased pitter-patter resistance of Cr–Mo steel welds. Acta Mater 2013;61(6):2194–206. link1
[18] Chen RP, Ghassemi Armaki H, Maruyama Chiliad, Igarashi Chiliad. Long-term microstructural degradation and pitter-patter strength in Gr.91 steel. Mater Sci Eng A 2011;528(13–14):4390–4. link1
[xix] Brinkman CR, Sikka VK, Horak JA, Santella ML. Long-term creep rupture beliefs of modified 9Cr–1Mo steel base and weldment behavior. Oak Ridge TN: Oak Ridge National Laboratory; 1987. Report No.: ORNL/TM-10504. link1
[20] Hald J. Microstructure and long-term creep backdrop of 9–12% Cr steels. Int J Press Vessels Piping 2008;85(1–2):30–7. link1
[21] Danielsen HK, Hald J. Behaviour of Z phase in 9–12% Cr steels. Energy Mater 2006;ane(1):49–57. link1
[22] Suzuki K, Kumai S, Kushima H, Kimura G, Abe F. Precipitation of Z-stage and precipitation sequence during pitter-patter deformation of modernistic. 9Cr–1Mo steel. Tetsu to Hagane 2003;89(vi):691–8. link1
[23] Danielsen HK. Z-phase in 9–12% Cr steels [dissertation]. Lyngby: Technical University of Kingdom of denmark; 2007. link1
[24] Cerri E, Evangelista E, Spigarelli South, Bianchi P. Evolution of microstructure in a modified 9Cr–1Mo steel during short term creep. Mater Sci Eng A 1998;245 (2):285–92. link1
[25] Smith DJ, Walker NS, Kimmins ST. Type IV creep cavity accumulation and failure in steel welds. Int J Press Vessels Pipage 2003;80(9):617–27. link1
[26] Lee JS, Maruyama G, Nonaka I, Ito T. Mechanism of blazon IV failure in weldment of a modern 9Cr–1Mo steel. In: Proceedings of the Creep Deformation and Fracture, Design and Life Extension; 2005 Sep 25–28; Pittsburgh, PA, U.s.a.; 2005. p. 139–48. link1
[27] Francis JA, Mazur W, Bhadeshia HKDH. Estimation of blazon IV keen tendency in ability institute steels. ISIJ Int 2004;44(xi):1966–8. link1
[28] Maziasz PJ, Klueh RL, Vitek JM. Helium furnishings on void germination in 9Cr– 1MoVNb and 12Cr–1MoVW irradiated in HFIR. J Nucl Mater 1986;141– 143:929–37. link1
[29] Klueh RL, Kai JJ, Alexander DJ. Microstructure-mechanical properties correlation of irradiated conventional and reduced-activation martensitic steels. J Nucl Mater 1995;225:175–86. link1
[30] Lilliputian EA, Stoter LP. 11th conference on ''furnishings of radiation on materials". Philadelphia: ASTM STP; 1982. link1
[31] Gelles DS, Thomas LE. Ferritic alloys for utilize in nuclear energy technologies. Warrendale: TMS-AIME; 1984. link1
[32] Orlova A, Buršik J, Kucharˇová K, Sklenicˇka V. Microstructural stability of pitter-patter resistant alloys for high temperature plant applications. London: The Institute of Materials; 1998. link1
[33] Laha K, Chandravathi KS, Parameswaran P, Bhanu Sankara Rao Thou, Mannan SL. Label of microstructures across the oestrus-affected zone of the modified 9Cr–1Mo weld joint to understand its role in promoting type 4 cracking. Metall Mater Trans A 2007;38(ane):58–68. link1
[34] Danielsen HK, Hald J. A thermodynamic model of the Z-phase Cr(V,Nb)N. Calphad 2007;31(4):505–14. link1
[35] Abe F. Analysis of creep rates of tempered martensitic 9%Cr steel based on microstructure development. Mater Sci Eng A 2009;510–511:64–ix. link1
[36] Parker JD, Coleman Chiliad, Henry J, Liu Westward, Zhou K. Guidelines and specifications for high-reliability fossil power plants: best practice guideline for manufacturing and construction of Grade 91 steel components 1023199. California: Electrical Ability Research Institute; 2011. Report No.: 3002006390.
[37] Foldyna V, Kubon Z, Vodarek V, Purmensky J. How to improve creep rupture forcefulness of avant-garde chromium steels. In: Proceedings of the Thrid International Briefing on Advances in Materials Technology for Fossil Power Plants; 2001 Apr 5–vi; Llandysul, Great britain; 2001.
[38] Klueh RL. Elevated temperature ferritic and martensitic steels and their application to future nuclear reactors. Int Mater Rev 2013;50(v):287–310. link1
[39] Viswanathan R, Nutting J. Advanced heat resistant steel for power generation. London: Institute of Materials; 1999. link1
[40] Zhong Y, Ozturk Thou, Sofo JO, Liu Z. Contribution of commencement-principles energetics to the Ca–Mg thermodynamic modeling. J Alloys Compd 2006;420(one–two):98–106. link1
[41] Asadikiya M, Rudolf C, Zhang C, Boesl B, Agarwal A, Zhong Y. Thermodynamic modeling and investigation of the oxygen effect on the sintering of B4C. J Alloys Compd 2017;699:1022–9. link1
[42] Asadikiya Thousand, Zhong Y. Oxygen ion mobility and conductivity prediction in cubic yttria-stabilized zirconia single crystals. J Mater Sci 2018;53(3): 1699–709. link1
[43] Costa e Silva A. Applications of multicomponent databases to the improvement of steel processing and design. J Phase Equilibria Diffus 2017;38(vi):916–27. link1
[44] Bale CW, Bélisle East, Chartrand P, Decterov SA, Eriksson Chiliad, Hack K, et al. FactSage thermochemical software and databases—recent developments. Calphad 2009;33(two):295–311. link1
[45] Andersson JO, Helander T, Höglund L, Shi P, Sundman B. Computational tools for materials science. Calphad 2002;26(2):273–312. link1
[46] Kroupa A. Modelling of phase diagrams and thermodynamic backdrop using Calphad method—evolution of thermodynamic databases. Comput Mater Sci 2013;66:3–13. link1
[47] Perrut M. Thermodynamic modeling by the Calphad method and its applications to innovative materials. Aerospace Lab 2015;9:1–11. link1
[48] Smith A, Asadikiya M, Zhong Y. The thermodynamic evaluation and modeling of Grade 91 alloy and its secondary phases through the CALPHAD arroyo. In: Proceedings of the 2017 Annual Review Meeting for Crosscutting Research MS&T; 2017 Mar xx–23; Pittsburgh, PA, Usa; 2017. link1
[49] Fedorova I, Kostka A, Tkachev Due east, Belyakov A, Kaibyshev R. Tempering behavior of a low nitrogen boron-added 9%Cr steel. Mater Sci Eng A 2016;662:443–55. link1
[fifty] Kipelova AY, Belyakov AN, Skorobogatykh VN, Shchenkova IA, Kaibyshev RO. Tempering-induced structural changes in steel 10Kh9K3V1M1FBR and their effect on the mechanical backdrop. Metal Sci Heat Care for 2010;52(3–4): 100–10. link1
[51] Kipelova A, Kaibyshev R, Belyakov A, Molodov D. Microstructure evolution in a 3%Co modified P911 heat resistant steel nether tempering and pitter-patter conditions. Mater Sci Eng A 2011;528(3):1280–6. link1
[52] Fedoseeva A, Dudova N, Kaibyshev R. Creep strength breakdown and microstructure evolution in a 3%Co modified P92 steel. Mater Sci Eng A 2016;654:1–12. link1
[53] Dudova N, Plotnikova A, Molodov D, Belyakov A, Kaibyshev R. Structural changes of tempered martensitic nine%Cr–2%Due west–three%Co steel during creep at 650 C. Mater Sci Eng A 2012;534:632–9. link1
[54] Cipolla L, Danielsen HK, Venditti D, Di Nunzio PE, Hald J, Somers MAJ. Conversion of MX nitrides to Z-phase in a martensitic 12% Cr steel. Acta Mater 2010;58(2):669–79. link1
[55] Laha Yard, Chandravathi KS, Parameswaran P, Rao KBS. Type Four cracking susceptibility in weld joints of unlike grades of Cr–Mo ferritic steel. Metall Mater Trans A 2009;xl(2):386–97. link1
[56] Sawada K, Tabuchi Thousand, Hongo H, Watanabe T, Kimura K. Z-phase formation in welded joints of high chromium ferritic steels after long-term creep. Mater Charact 2008;59(nine):1161–7. link1
dalrympleyoungold.blogspot.com
Source: https://www.engineering.org.cn/en/10.1016/j.eng.2019.12.004
0 Response to "Review of Type Iv Cracking in Piping Welds Ellis Viswanathan"
Post a Comment